Zeroplus 02392 Manual de usuario

02392
EN Operating Instructions
from page 3
02392_EN_180x140_GM_V1.indd 102392_EN_180x140_GM_V1.indd 1 19.11.2019 15:54:1319.11.2019 15:54:13

02392_EN_180x140_GM_V1.indd 202392_EN_180x140_GM_V1.indd 2 19.11.2019 15:54:1619.11.2019 15:54:16

3 EN
Contents
Explanation of the Symbols _______________ 4
Explanation of the Signal Words____________ 5
Intended Use ___________________________ 5
Indications _____________________________ 5
Contraindications _______________________ 6
Read Carefully Before First Use ____________ 8
Safety Notices __________________________ 8
Product Description_____________________ 12
Before Initial Use_______________________ 12
Items Supplied_________________________ 13
Device Description _____________________ 13
Operation ____________________________ 14
Cleaning and Storage ___________________ 16
FAQs ________________________________ 17
Technical Data ________________________ 18
Disposal______________________________ 19
Safety and Performance Standards ________ 19
Dear Customer,
We are delighted that you have chosen this ultrasound
pain therapy device.
Before using this device for the first time, please read
the operating instructions carefully and keep them for fu-
ture reference. The operating instructions form an inte-
gral part of the device. If the device is passed on to oth-
ers, these operating instructions must also be passed
on together with the device.
The manufacturer and importer do not accept any liabili-
ty if the information in these operating instructions is not
complied with.
If you have any questions about the device and about
spare parts / accessories, contact the customer service
department via our website:
www.dspro.de/kundenservice
02392_EN_180x140_GM_V1.indd 302392_EN_180x140_GM_V1.indd 3 19.11.2019 15:54:1619.11.2019 15:54:16
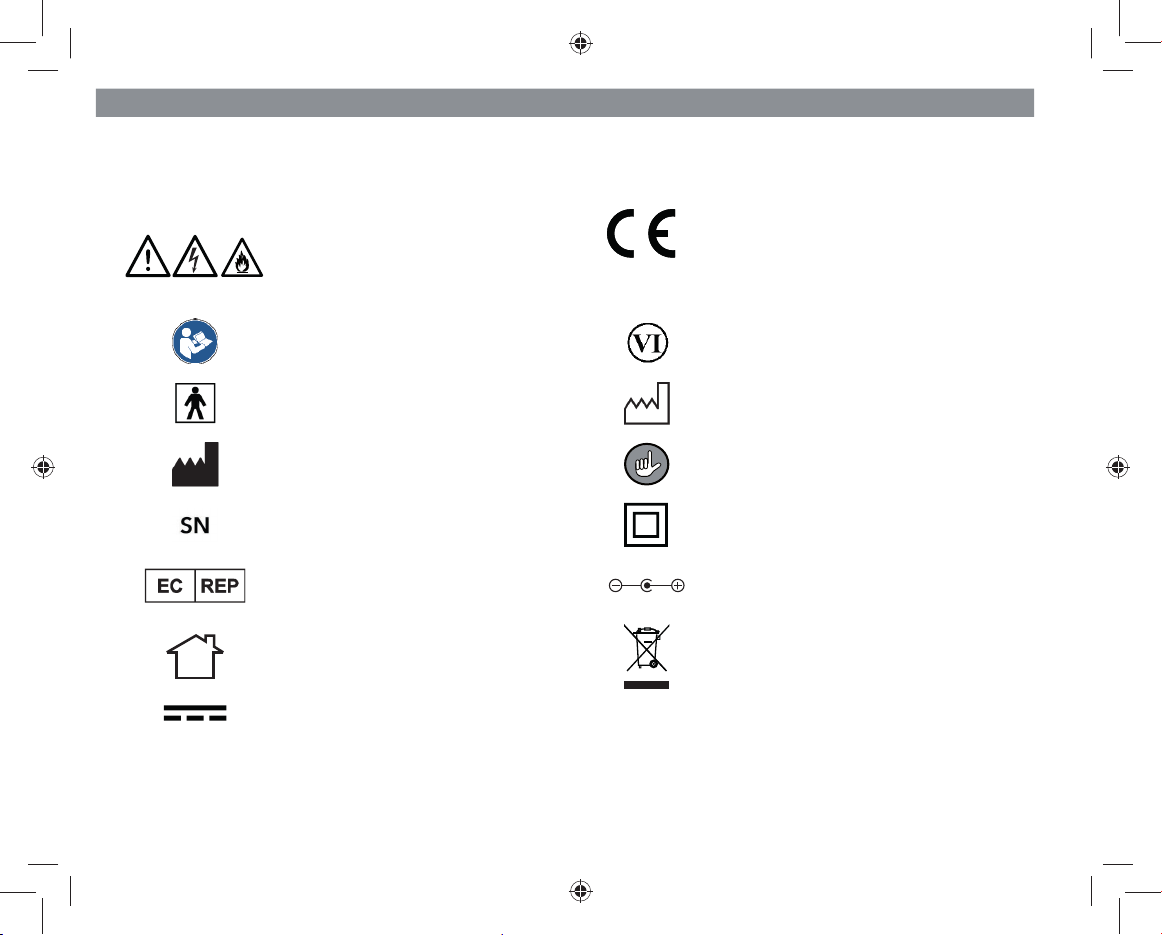
EN 4
Explanation of the Symbols
The following symbols are used in the operating instructions for the “ultrasound pain therapy device” or accessories:
Danger symbols: These symbols
indicate dangers of injury. Read
through the associated safety notic-
es carefully and follow them. 0123
Concordance with Directive 93/42
EEC
Read the operating instructions
before use Energy efficiency class VI
Type BF applied part Date of manufacture
Manufacturer Supplementary information
Specification of the serial number Protection class II device
EU Representative Polarity of the coaxial connector
(mains adapter)
Only use indoors!
Electrical and electronic devices must
be properly disposed of in accord-
ance with the WEEE Directive
Direct current IP22 Protection type
02392_EN_180x140_GM_V1.indd 402392_EN_180x140_GM_V1.indd 4 19.11.2019 15:54:1619.11.2019 15:54:16

5 EN
Explanation of the Signal Words
DANGER Warning of serious and life-threatening injuries.
WARNING Warning of possible serious and life-threatening injuries.
NOTICE Warning of damage to property.
Intended Use
• The device is intended to be used only on humans. Use the device exclusively for the applications described.
• This device is intended to be used for medical care at home.
• The device is designed exclusively for personal use and must not be used for commercial purposes.
• The device does not require any calibration, preventive inspection or maintenance measures.
• The device is not equipped with any parts that need to be maintained by the user.
• The device is protected against unauthorised use. Never modify the device without the manufacturer's approval.
If you modify the device, suitable inspection and test procedures must be carried out in order to ensure that the
device is still safe for use.
• All defects caused by improper handling or damage are excluded from the warranty. The same applies to normal
wear and tear.
Indications
• Pain relief, muscle spasms and joint contractures
• Symptomatic relief of chronic persistent pain
• Post-traumatic and post-operative pain
02392_EN_180x140_GM_V1.indd 502392_EN_180x140_GM_V1.indd 5 19.11.2019 15:54:1719.11.2019 15:54:17

EN 6
Contraindications
If you have any health concerns, suffer from health problems or belong to a risk group, consult your
doctor before using the device.
• The following people must not use the device or do so only after consulting a doctor:
–people who wear a pacemaker or similar medical implants (e.g. a neurostimulation device);
–people with heart disease or who suffer from epilepsy;
–women who are pregnant, have just given birth or are breastfeeding;
–people who suffer from infectious diseases or circulation problems;
–people with incomplete skeletal growth;
–people who have undergone an operation for which the treatment is not yet complete;
–people with carcinogenic lesions;
–people with malignant tumours;
–people with haemorrhagic diathesis (strong bleeding propensity);
–people with local inflammation;
–people with tuberculosis;
–people with septic inflammation;
–people with diabetes mellitus;
–people with osteoporosis;
–people who have had a laminectomy;
–people with an endoprosthesis / metal implant;
–people with a thrombophlebitis and / or varicose veins;
–patients with a fever (pyrexia).
02392_EN_180x140_GM_V1.indd 602392_EN_180x140_GM_V1.indd 6 19.11.2019 15:54:1719.11.2019 15:54:17

7 EN
• Do not use the device near to or on the following areas:
–heart;
–chest area for people who wear a pacemaker or similar medical implants;
–brain tissue;
–carotid sinus nerves;
–carotid artery;
–laryngeal or throat muscles;
–after undergoing a laminectomy not over the affected area of the spine;
–a healing fracture;
–body parts with no feeling;
–body parts with post-traumatic consequential damages;
–numb body parts;
–ossification centres (in the case of incomplete bone growth);
–acute injuries and open wounds;
–benign or malignant tumours;
–eyes;
–uterus;
–testicles;
–genital area.
• This device should not be used to alleviate symptomatic local pain, unless the cause of the illness is known or a
pain syndrome has been diagnosed.
• The other contraindications include patients who are suspected to have illnesses for which it is advisable to avoid
heat and fever for general medical reasons.
• If you experience pain or skin irritation during or after use of the device, stop using the device immediately and
consult a doctor before using the device again.
02392_EN_180x140_GM_V1.indd 702392_EN_180x140_GM_V1.indd 7 19.11.2019 15:54:1719.11.2019 15:54:17

EN 8
Read Carefully Before First Use
Caution!
• Read, understand and follow the safety precautions and these operating instructions. You should be
aware of the restrictions and risks which occur when using all ultrasound devices.
• Regularly find out information about all contraindications.
• The ultrasound device should be subjected to a routine check before each use to check whether all oper-
ating elements are working properly and in particular whether the intensity regulation adjusts the intensity
of the ultrasound power that is emitted stably and correctly. You should likewise check whether the reg-
ulation of the treatment time actually ends the emission of the ultrasonic waves when the timer runs out.
Check: Hold the device horizontally with the ultrasound head pointing upwards. Place a drop of water in
the middle of the ultrasound head and switch the device on (see the “Operation” chapter). If the device
functions correctly, the drop of water will move around the surface area of the ultrasound head or it might
even disperse.
Safety Notices
■This device may be used by people with reduced physical, sensory or mental abilities
or a lack of experience and / or knowledge if they are supervised or have received in-
struction on how to use the device safely and have understood the dangers resulting
from failure to comply with the relevant safety precautions.
■The device may not be used by children. Children and animals must be kept away
from the device and the connecting cable.
■Close supervision is required if the device is used by or in the vicinity of children or
fragile people.
■Children should be supervised to ensure that they do not play with the device.
No particular training is required to use this device.
■The device should only be used and stored out of the reach of children and animals.
02392_EN_180x140_GM_V1.indd 802392_EN_180x140_GM_V1.indd 8 19.11.2019 15:54:1719.11.2019 15:54:17

9 EN
■The device must always be disconnected from the mains power when left unattended
and before cleaning it.
■The mains adapter contains a transformer. Do not replace the mains adapter with
another adapter as this can be dangerous.
■In the interest of your own safety, check the device and the connecting cable for
damage before each use. Only use the device if it is in good working order. If the
connecting cable or device are damaged, contact the customer service department
or your dealer as repairs can only be carried out with a special tool.
■Do not use a damaged device.
■Never try to repair the device yourself. This could cause serious injuries. Failure to
follow this specification will cause the warranty to become void.
DANGER – Danger of Electric Shock
■Use the device only in closed rooms.
■Do not use the device in a wet environment (e.g. in a bathroom or near to a shower). Never immerse the device
in water or other liquids.
■Penetration of liquids into the device should be prevented as this can cause a malfunction of the internal parts and
thus entail a risk of injury to the user.
■Should the device fall into water, switch offthe power supply immediately. Do not attempt to pull the device out of
the water while it is still connected to the mains power!
■Never touch the device and the connecting cable with wet hands when the device is connected to the mains
power.
02392_EN_180x140_GM_V1.indd 902392_EN_180x140_GM_V1.indd 9 19.11.2019 15:54:1719.11.2019 15:54:17

EN 10
WARNING – Danger of Injury
■Danger of injury and suffocation! Keep children and animals away from the device and packaging material.
■Danger of strangulation! Make sure that the connecting cable is always kept out of the reach of small children
and animals.
■Danger of burns from heated ultrasound head!
–Make sure that you do not hold the ultrasound head on one spot for too long. It must constantly be kept moving.
–Make sure that you use a sufficient amount of ultrasonic gel.
–The recommended length of use is no more than 15 minutes. Do not exceed this!
–Allow the device to cool down for approx. 30 minutes after use before using it again.
–Do not leave the device switched on without contact with the skin. There is a danger of overheating!
■Operating the device in a way other than the one described here or failure to follow the operating steps described
here may cause dangerous ultrasound radiation to be released.
■The device must not be operated in the vicinity (i.e. within a radius of less than 1 metre) of short-wave or micro-
wave ovens.
■Do not use a mobile phone while using the device.
■Do not use the device while you are connected to other medical devices.
■Do not use the device in an environment in which devices which deliberately release electromagnetic radiation
without a shield are used.
■Disinfect the ultrasound head with a cloth soaked in alcohol before the device is used by another person. This
prevents any transfer of skin infections or similar complaints.
■Danger of tripping! Lay the connecting cable in such a way that it does not present a trip hazard.
02392_EN_180x140_GM_V1.indd 1002392_EN_180x140_GM_V1.indd 10 19.11.2019 15:54:1819.11.2019 15:54:18
Tabla de contenidos
Manuales populares de Terapia de luz de otras marcas

Lumie
Lumie Vitamin L Manual de usuario

Philips
Philips Pro HF3331/60 Manual de usuario

iReliev
iReliev ET-1616 Manual de lista de piezas

DEMYK LIGHTMOD PRODUCTS
DEMYK LIGHTMOD PRODUCTS SENSOSPHERE-Learning Aid Manual de usuario

Beurer
Beurer TL 85 Manual de usuario

Philips
Philips GoLite HF3332 Manual de usuario











