Micro Direct MicroLab Manual de usuario

MicroLab
Operating Manual
Federal (USA) law restricts this device to sale by or on
the order of a physician or licensed practitioner.
MAN1300
085-73
Issue 1.6
February 2019
Micro Direct, Inc.
803 Webster Street
Lewiston, ME 04240
1-800-588-3381
www.mdspiro.com
Mail In Yellow Warranty Card
Receive 25 PFT Filters
FREE!!

Indications for Spirometry
Spirometry has been used extensively to measure lung function capability and to
recognize and treat many diseases associated with the impairment of healthy lung
functions. Spirometry today provides great insight into the status of any person’s
health.
Generally speaking, spirometry is a simple diagnostic tool used to define a subject’s
lung condition. The major indications for spirometry are:
✓Dyspnea (shortness of breath)
✓Exercise induced coughing
✓Chest tightness
✓Smokers over 45 years of age (NLHEP recommendations)
✓Obesity
✓Pre-operative testing
✓Occupational exposure to dust and/or chemicals
✓Ongoing assessment of patients receiving bronchodilator treatments
✓Determination and/or documentation of pulmonary disability
✓Asthma diagnosis
✓Pre-existing pulmonary disease
✓Frequent colds
✓Assessment of congestive heart failure
CPT Codes for Spirometry
94010 - Spirometry Complete
Includes graphic record total and timed vital capacity, expiratory flow rate
measurement (s) with or without maximal voluntary ventilation
94060 - Bronchodilation Responsiveness
Spirometry as in 94010, pre-and post-bronchodilator or exercise
94070 - Bronchospasm Provocation Evaluation
Multiple spirometric determinations after bronchodilator with spirometry as in
94010
94150 - Vital Capacity
Total (separate procedure)
94200 - Maximal Voluntary Ventilation
Maximum breath capacity
94375 - Flow Volume Loop
Respiratory Flow Volume Loop
95070 - Inhalation Bronchial Challenge Testing
(Not including necessary pulmonary function tests), with histamine, methacholine
or similar compounds.
94464 - Bronchodilator Administration
Demonstration and/or evaluation of patient utilization of an aerosol generator,
nebulizer and meter dose inhaler or IPPB device
Diagnosis and ICD-10-CM Codes on back cover

Contents
Introduction.........................................................................1
Contraindications................................................................1
Warning and Cautions........................................................2
Indication for Use................................................................2
Overview ............................................................................4
Getting Started ...................................................................5
Calibration Check (Verification).........................................14
Customization...................................................................17
Administration Mode.........................................................18
Paper Loading..................................................................19
Switching Off ....................................................................20
Charging Procedure..........................................................20
PC connection using SPCS..............................................20
Looking after your Spirometer...........................................21
Product Lifetime................................................................21
Cleaning Instructions........................................................21
External Surfaces of the Spirometer.................................21
Cleaning the Accessories .................................................22
Cleaning the Transducer...................................................22
Servicing...........................................................................23
Troubleshooting Information.............................................24
Safety Designation per IEC 60601-1.................................26
Electromagnetic Compatibility (EMC) to IEC 60601-1-2....27
Symbols............................................................................31
Specifications ...................................................................32
Spirometry Measurements................................................32
Consumables / Supporting Products.................................34
ICD-10 Codes for Spirometry............................................35

1
Introduction
The MicroLab is a mains/battery operated portable spirometer with the
unique combination of ease of use and sophistication. Ease of use is
assured using context sensitive help screens, accessed at a touch of a
button, that explain every MicroLab feature.
The MicroLab uses a Digital Volume Transducer, an extremely stable
form of volume transducer, which measures expired air directly at B.T.P.S
(Body Temperature and Pressure with Saturated water vapor) thus
avoiding the inaccuracies of temperature corrections. The transducer is
insensitive to the effects of condensation and temperature and avoids the
need for individual calibration prior to performing a test
Test results may be uploaded to a PC using the optional Spirometry PC
Software and patient details may be downloaded to the MicroLab.
Stored data may be printed to the integral thermal or uploaded to a PC
using the optional Spirometry PC Software (SPCS).
Contraindications
Contraindications: It is recommended that patients should
not be tested within one month of a myocardial infarction.
Conditions where suboptimal spirometry are likely:
•chest or abdominal pain
•oral or facial pain exacerbated by a mouthpiece
•stress incontinence
•dementia or confused state
Ref: ATS/ERS Task Force: Standardization of Lung
Function Testing. General considerations for lung function
testing.
M. Miller et al. Eur Resp J 2005:26. 153-161

2
Warning and Cautions
The following terms are used as follows in this manual
CAUTION: Possibility of injury or serious damage
WARNING: Conditions or practices that could result in personal injury
Note: Important information for avoiding damage to the instrument or
facilitating operation of the instrument.
Note: Patients below the age of four (4) may struggle to perform
spirometry correctly and reproducibly.
Note: The device should be used by trained and qualified personnel.
Indication for Use
The MicroLab spirometer is intended, for prescription use only, to
measure the maximal volume and flow of air that can be moved in and
out of a patient's lungs.
The system is intended for use with pediatric (4 to 17 years) and adult (18
to 99 years) patients in hospitals, physician offices, laboratories and
occupational health testing environments.

3
CAUTION: Read the manual before use.
WARNING: The instrument is not suitable for use in the presence of
explosive or flammable gases, flammable anesthetic mixtures or in
oxygen rich environments.
CAUTION: Mouthpieces are single patient use. If used on more than one
patient, there is a risk of cross-infection. Repeat use may degrade
materials and lead to an incorrect measurement.
CAUTION: Pulmonary filters are single patient use. If used on more than
one patient, there is a risk of cross-infection. Repeat use may increase air
resistance and lead to an incorrect measurement.
PLEASE NOTE: The product you have purchased should not
be disposed of as unsorted waste. Please utilize your local
recycling facility for the disposal of this product.
PLEASE NOTE: Degree of protection against Ingress of Water is IPX0.
WARNING: To avoid risk of electric shock, this equipment must only be
connected to a supply mains with protective earth.
WARNING: Do not connect devices that are not specified as part of the
system.

4
Overview
The MicroLab uses a touch screen
with icons representing each function
available. A stylus, housed in the left-
hand side of the unit, is provided for
icon screen activation.
Touch the displayed time to adjust
time and date. Touch the toolbox icon
to adjust volume and brightness.
Unused icons may be disabled by
touching the blue background and
selecting from the list displayed.
Four levels of battery charge are
indicated by the segmented battery
icon. When this icon turns red the
battery is nearly exhausted and the
batteries must be charged –see
Charging Procedure.
.
The complete functionality is described
on the help screen.
This is obtained by pressing the help
button (?).
Help text exists for every screen
viewed during the operation of the
MicroLab.
You are recommended to make full
use of the extensive Help screens
provided.

5
Getting Started
When performing a spirometry
test, the recommended workflow
is to enter the patient’s details, or
retrieve them from memory,
perform the required test and
then print and save the results.
Please ensure that the turbine
transducer is plugged in to either
of the two sockets on the right-
hand side of the instrument.
6
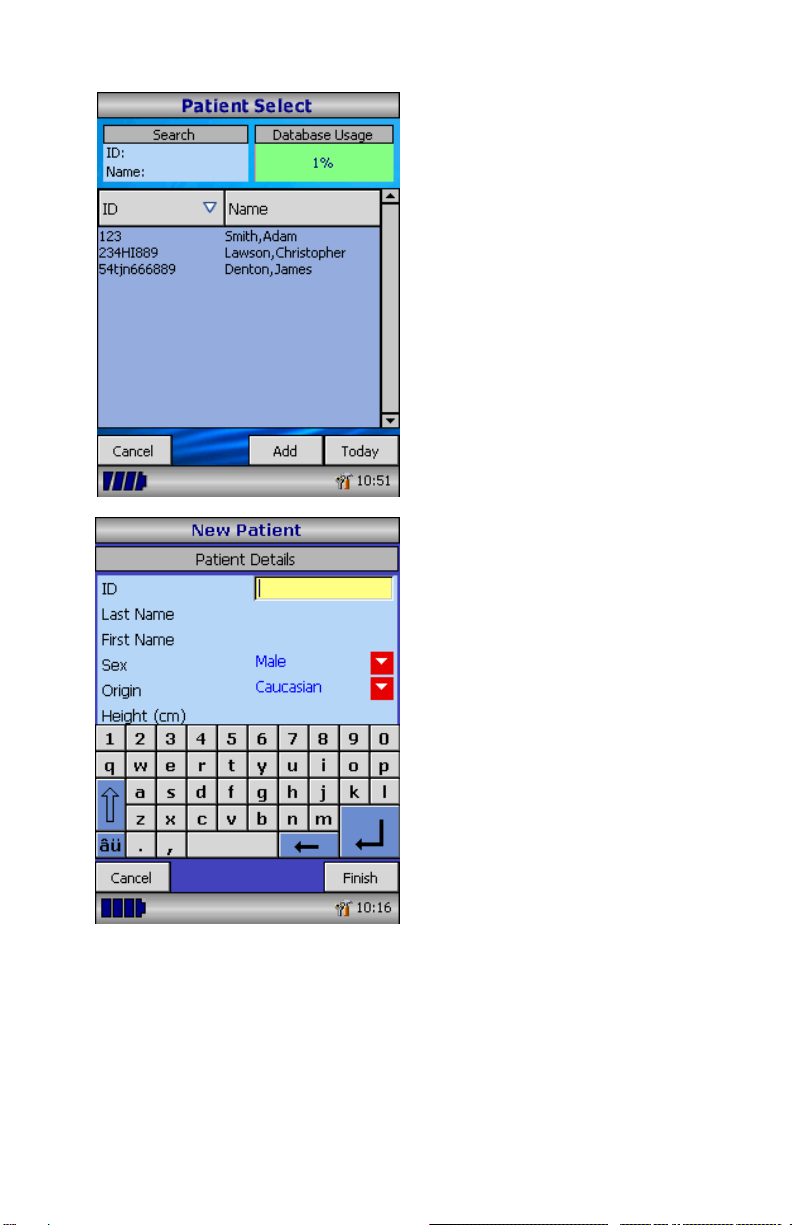
Select the ‘Patients’ icon to enter the
patient database.
The required patient may be selected
from the stored patient list.
If the patient details have not been
previously stored, select ‘Add’ to
enter the new patient’s details. The
patient details may also be
downloaded from the optional
Spirometry PC Software.
Once selected, the patient’s name
will appear at the bottom of the
screen.
Use the help button to obtain further
information.
To add a patient to the database, use
the on-screen keyboard to type a
unique patient ID and then touch the
enter key.
You will then be prompted for Last
Name, First Name, Sex, Ethnic
Origin, Height, Weight, date of Birth
and Factor.
A factor can be applied when testing
individuals of other ethnic origins who
would not normally be tested against
the countries set of predicted values.
The factor alters the predicted value
set on volume indices by the
percentage applied. If NHANES
predicted values are selected, then
the ethnic origin field should be
chosen but a factor correction is not
required.

7
The following factors are recommended when using ECCS normal
values:
Hong Kong Chinese 100%
Japanese American 89%
Polynesians 90%
North Indians and Pakistanis 90%
South Indians and those of African descent 87%
Ref: Lung Volumes and Forced Ventilatory Flows. P.H. Quanjet et al. Eur
Respir J, 1993, 6, Suppl. 16p5-40.
Once all the patient details are
added, the patient is added to the
database and the main menu is
displayed with the patient name
displayed at the bottom of the screen.
From the main menu select the
required test, by touching the icon
with the stylus.
If the displayed patient is not required
for testing, touch the patients name
and options to change or remove the
current patient will become available.
Tabla de contenidos
Otros manuales de Equipo médico de Micro Direct




















